Next: NPH and NPT MD Up: Molecular Dynamics at Constant Previous: Liouville Equation
![]()
![]()
![]()
Next: NPH and NPT MD
Up: Molecular Dynamics at Constant
Previous: Liouville Equation
How can we make a transition from the constant energy to the constant temperature? By coupling to the environment, or to the thermal bath.
Coupling to the environment is simulated by random ``collisions'' with imaginary heat bath particles. These collisions lead to instantaneous momentum changes. The particle momenta are reset to new values, taken from the Maxwell distribution. This way the average kinetic energy is always right.
The natural variation on this theme is resetting velocities of all particles at the same time after certain interval. Then the dynamics during this interval is truly microcanonical, and time correlation functions can be calculated inside this interval.
After the new velocities are assigned, the new configuration is accepted or rejected based on Metropolis-like criteria for Monte Carlo simulations. This technique was first suggested by Heyes in 1980, and then reinvented by D. Heermann et. al. in 1990 as Hybrid Monte Carlo. D. Heermann et. al. also showed, that this acceptance criterium is needed to account for the numerical integration errors. Otherwise this technique reproduces canonical ensemble only approximately.
An alternative way to simulate constant temperature is to rescale all the velocities to keep kinetic energy constant. It is a very crude approach used in the early days. If done on every step, it alters the system dynamics, which does not even correspond to the canonical ensemble (Why?). If done at certain intervals, it adds some periodic perturbation to the system, which is in general undesirable, but sometimes can serve as a tool to study system dynamics. Was used for simulations of glasses by Rahman et. al. in early 1980's. It is also often used to equilibrate the system during the the first few hundred MD steps before the production run starts and data are collected.
A more gentle and more practical way, known as
van Gunstern-Berendsen thermostat is to use a factor, that
depends on the deviation of the instantaneous kinetic energy K from
the average value K0, corresponding to desired temperature T0.
At each time step velocities are scaled by the factor ![]() :
:
| (7) |
Although this method does not reproduce canonical ensemble, it is widely used, and usually gives the same results as the rigorous method discussed below, although the caution must be taken using it. It reproduces the correct average energy, but the distribution is wrong. Therefore, averages are usually correct, but fluctuations are not.
This method does reproduce canonical ensemble, even though it also involves velocity rescaling.
The idea is to introduce an additional degree of freedom ![]() ,describing the external bath and corresponding velocity
,describing the external bath and corresponding velocity
![]() . Additional kinetic
. Additional kinetic ![]() , and
potential
, and
potential ![]() energy terms, coupled to the particles momenta, are added to the
Hamiltonian (Hoover, 1985). Using Hamilton equations (4) we
obtain equations of motion:
energy terms, coupled to the particles momenta, are added to the
Hamiltonian (Hoover, 1985). Using Hamilton equations (4) we
obtain equations of motion:
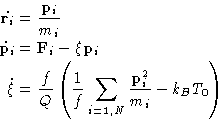 |
(8) |
![\begin{displaymath}
\rho \sim \exp\left[-\beta \left(\sum_{i=1,N}\frac{\mathbf{p}_i^2}{2m_i}+U({\mathbf{q}_1\dots\mathbf{q}_N})\right)\right]\end{displaymath}](img23.gif) |
(9) |
There is an alternative, but very similar method, called Nose thermostat, which also uses an additional variable for the momenta rescaling. It was introduced earlier (Nose, 1984). It works in the scaled coordinates, and reproduces canonical distribution for positions and scaled momenta.
© 1997 Boris Veytsman and Michael Kotelyanskii